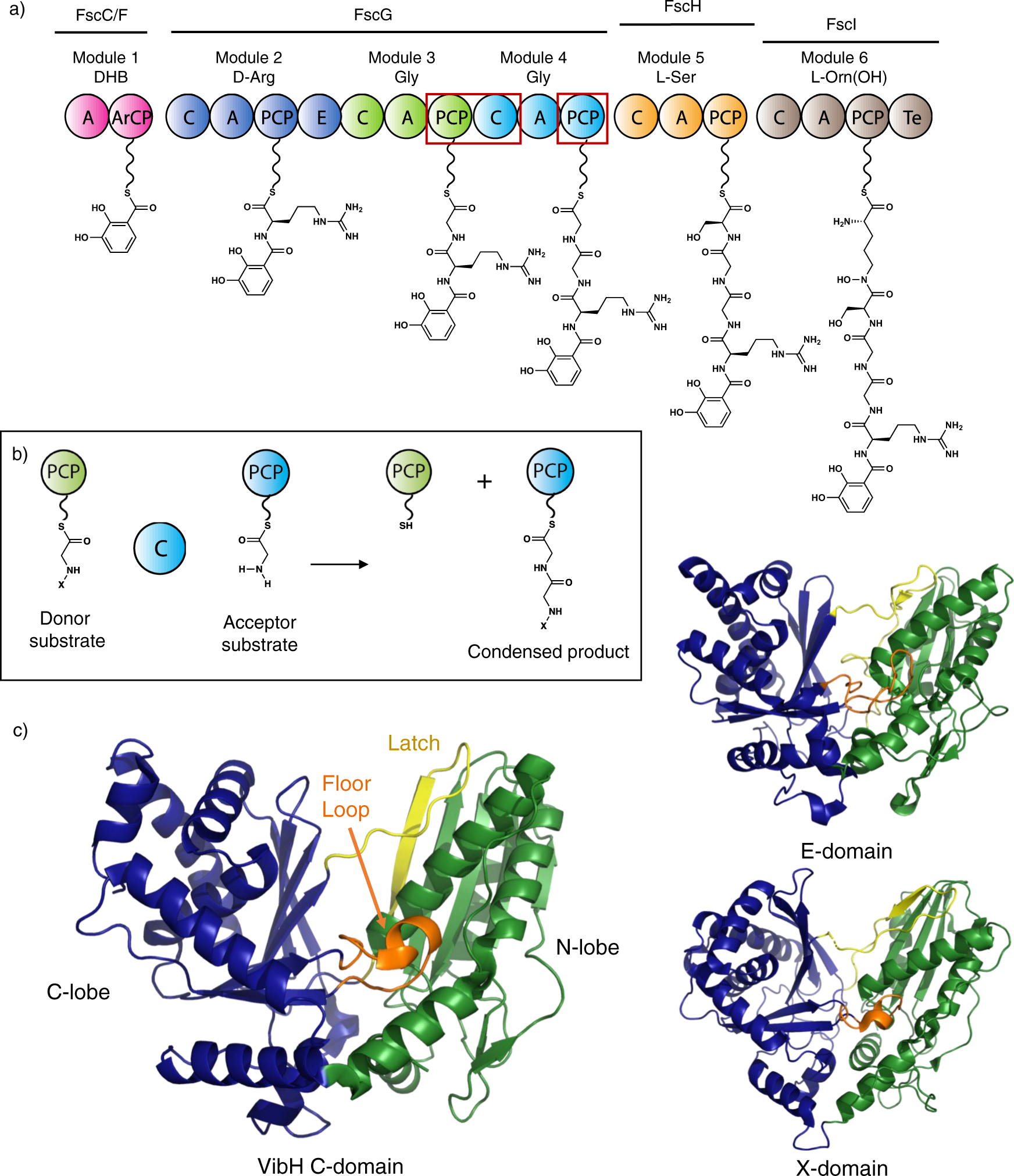
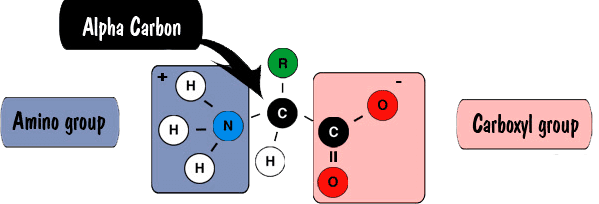
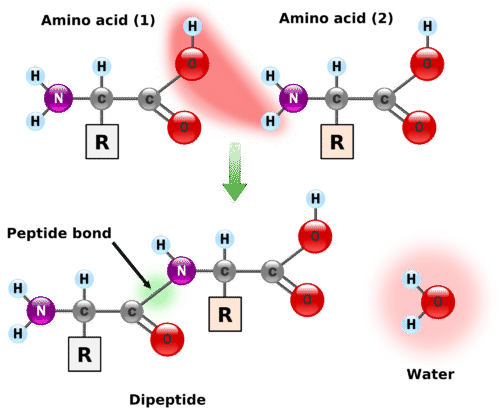
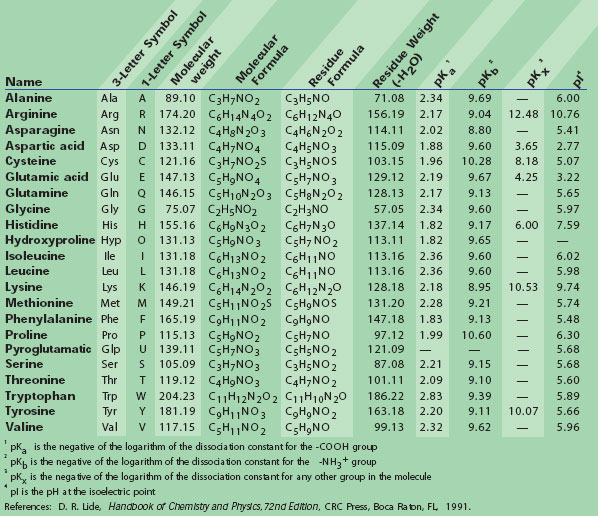
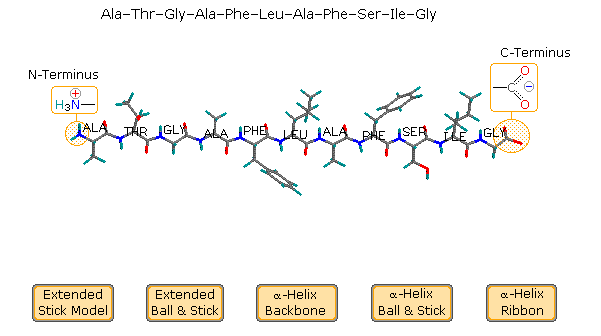
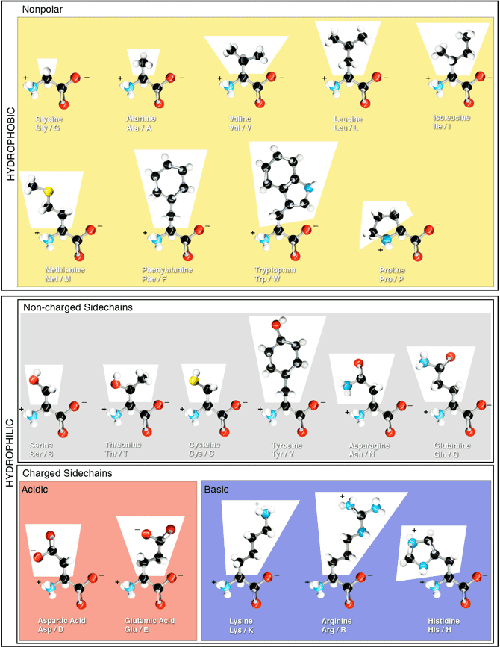
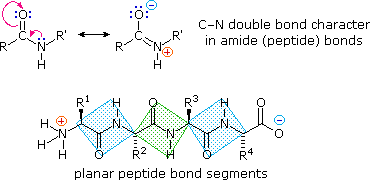
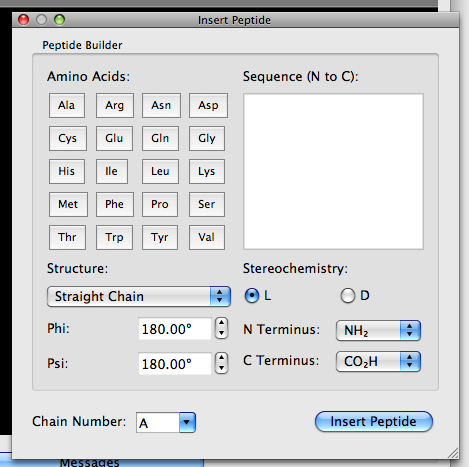
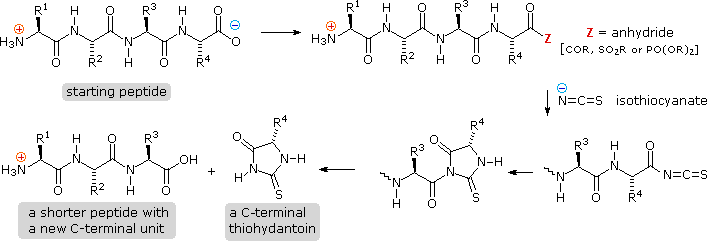
Structural Basis of Proline-Proline Peptide Bond Specificity of the Metalloprotease Zmp1 Implicated in Motility of Clostridium difficile - ScienceDirect
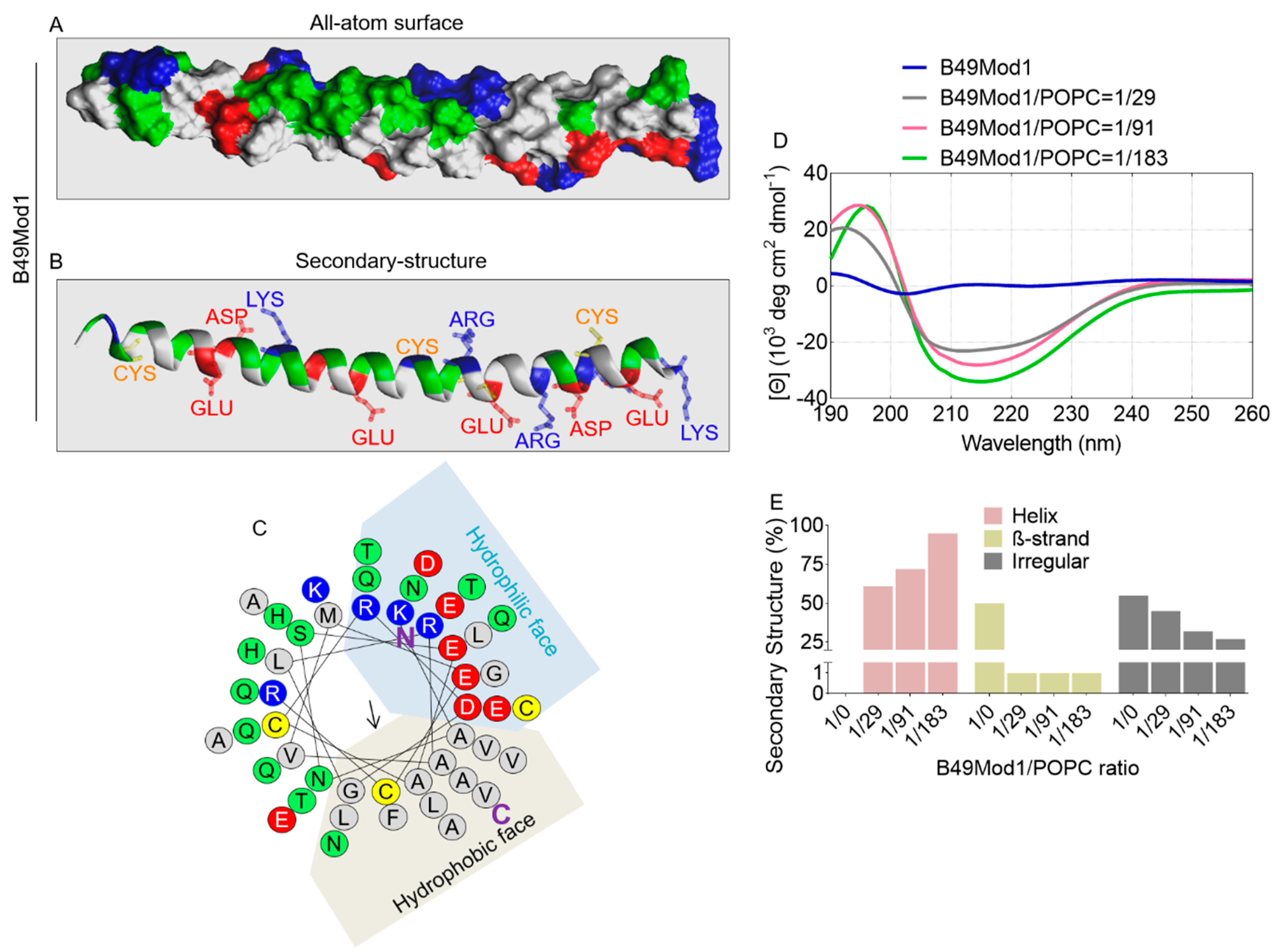
Molecules | Free Full-Text | Development and Characterization of the Shortest Anti-Adhesion Peptide Analogue of B49Mod1 | HTML
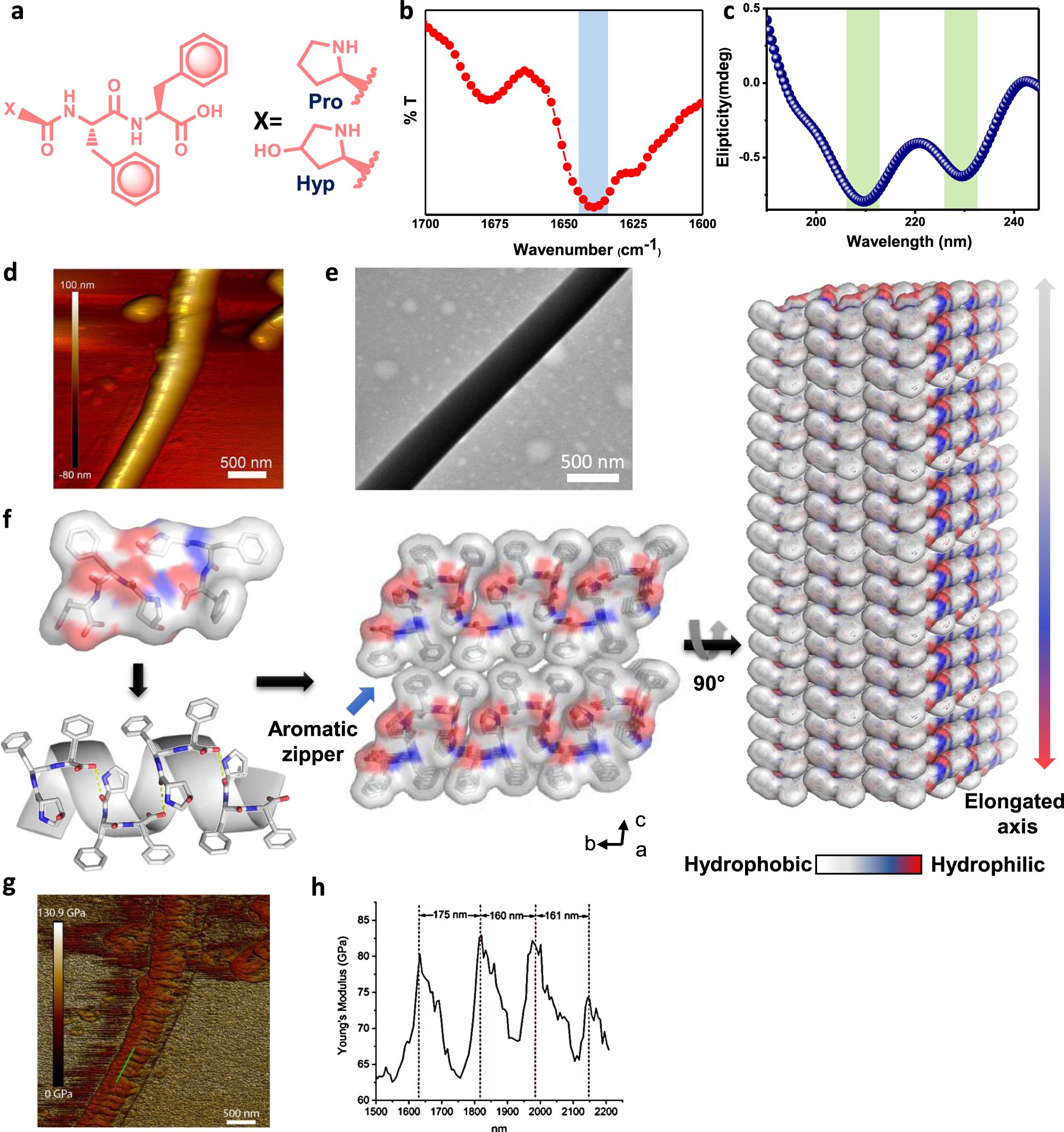
Molecular engineering of piezoelectricity in collagen-mimicking peptide assemblies | Nature Communications
CycloPs: Generating Virtual Libraries of Cyclized and Constrained Peptides Including Nonnatural Amino Acids